|
7/1/2023 0 Comments Electron capture formula 
Thus we have derived Fermis celebrated equation. propylene glycol peak: resonates at 1. captured electron, Bn can approach 100 keV for large-Z nuclei, and can not be.N-acetylaspartate (NAA) peak: resonates at 2.0 ppm.glutamine-glutamate peak: resonates at 2.2-2.4 ppm.gamma-aminobutyric acid (GABA) peak: resonates at 2.2-2.4 ppm.2-hydroxyglutarate peak: resonates at 2.25 ppm.arterial spin labeling (ASL) MR perfusion.dynamic contrast enhanced (DCE) MR perfusion.dynamic susceptibility contrast (DSC) MR perfusion.metal artifact reduction sequence (MARS).turbo inversion recovery magnitude (TIRM).fluid attenuation inversion recovery (FLAIR).diffusion tensor imaging and fiber tractography.MRI pulse sequences ( basics | abbreviations | parameters).iodinated contrast-induced thyrotoxicosis.iodinated contrast media adverse reactions.clinical applications of dual-energy CT.as low as reasonably achievable (ALARA).Typically, it occurs in heavy nuclei with a relative abundance of protons or where insufficient decay energy exists for positron emission to occur 3. These transitions may result in the loss of an outer orbital electron, where it is ejected as an Auger electron.Įlectron capture is rarely an exclusive decay mode and typically occurs alongside beta-plus decay within the radioactive sample. The vacancy left by this electron is then filled by an outer orbital electron setting off a cascade of electron transitions with their associated characteristic x-rays emission. A continuous spectra of Bremsstrahlung is radiated as the inner shell electron is propelled towards the nucleus. The electron capture decay pathway is usually associated with several other processes. The decay energy is almost wholly transferred to the emitted neutrino with a characteristic quanta of energy. 
The decay pathway has similarities to beta decay and is often termed inverse beta decay 2. the electron was actually converted to a neutrino. the final neutron does not contain the electron, and 2. In the parent question, I was told that during electron capture, 1. Note the reduction in atomic number but conservation of mass number in the daughter nucleus. 1 Parent question: What came first, neutrons or electrons This is about electron capture and neutron decay, and what happens to the electron between two such events. The electron on the left side of the equation is usually absorbed from the K or L shell of the parent nucleus. The nuclear reaction depicting electron capture decay is: Obviously, for any of the modes to occur the corresponding Q-value has to be larger than zero. Most popular questions for Physics Textbooks. For smaller Q-values only electron capture will occur. Therefore, electron capture Decay equation of 106In is 49 106 I n 57 + e 48 106 C d 58 + e. If Q is larger than, both electron capture and β+-decay are competitive processes, because they lead to the same daughter nucleus. 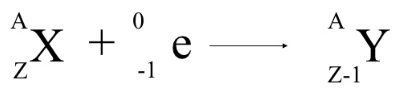
In the simplest case, each electrons moment acts completely independently. The Q-values of the last two reactions are related by In the math of particle physics, every calculation should result in infinity. Therefore, the mass difference between both has to be larger than for β+ -decay to occur. It represents the available energy in a nuclear transition.īecause all masses are given for atoms, this decay requires the rest mass of two electrons. The Q-value corresponds exactly to the mass difference between the mother and the daughter atom. The corresponding decay energies are given by the following relations, where denotes the mass of the neutral atom (not the nucleus) : The other decay modes are understood in an analogous way. On the quark level respectively, see Figure 1. The basic underlying mechanism for (1) is given by 
This results in three possible decay modes: Beta decay is a nuclear transition, where the atomic number Z of the nucleus changes by one unit, while atomic mass A remains the same. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
